Aberrant Expression of Lymphoid Antigen in the Patients with Acute Myeloid leukemia in Aleppo Governorate
هدفت الدراسة الحاليَّة إلى تقيَّيم انتشار حالات التعبير المناعي الشاذ عن مستضدات الخلايا اللمفاويَّة لدى المرضى المصابين بالابيضاضات النقويَّة الحادة في محافظة حلَّب بالاعتماد على تقانة التنميط المناعي بالتدفق الخلويَّة خلال الفترة الممتدة من كانون الثاني ولغاية كانون الأول لعام 2017. شملت هذه الدراسة 72 حالة لمرضى شُخِصَ لديهم الإصابة بالابيضاضات النقويَّة الحادة Acute myeloid leukemia (AML).
مجلة بحوث جامعة حلبAberrant Expression of Natural Killer Cell Antigen in the Patients with Acute Myeloid leukemia in Aleppo Governorate
هدفت الدراسة الحاليَّة إلى تقيَّيم انتشار حالات التعبير المناعي الشاذ عن المستضدات الخاصة بالخلايا القاتلة طبيعياًCells Natural Killer (NK Cell) لدى المرضى المصابين بالابيضاضات النقويَّة الحادة في محافظة حلب بالاعتماد على تقانة التنميط المناعي بالتدفق الخلوي، شَمِلت الدراسة الحاليَّة 147 حالة مرضيَّة لمرضى شُخِصَ لديهم الإصابة بالابيضاضات الدمويَّة الحادة، تضمنت 72 حالة من النمط النقوي الحاد و70 حالة من النمط اللمفاوي الحاد، و5 حالات من النمط الظاهري المختلط. توزع المرضى المصابين بالابيضاضات النقويَّة الحادة إلى 46 حالة من الذكور و26 حالة من الإناث بنسبة (1:1.7).
مجلة بحوث جامعة حلبAberrant Expression of Myeloid Antigen CD13 and CD33 in the Patients with Acute Lymphoblastic leukemia in Aleppo Governorate
هدفت الدراسة الحالية إلى تقييم انتشار التعبير المناعي الشاذ عن المستضدات النقوية CD13 وCD33 لدى المرضى المصابين بالابيضاضات اللمفاوية الحادة في محافظة حلب بالاعتماد على تقانة التنميط المناعي بالتدفق الخلوي، شملت الدراسة الحالية 141 حالة مرضية لمرضى شُخِصَ لديهم الإصابة بالابيضاضات الدموية الحادة. وقد اشارت نتائج الدراسة الحالية لتوزع المرضى كالتالي، 67 حالة من المرضى شُخِصَ لديهم الإصابة بالابيضاضات النقوية الحادة AML بنسبة (47.5%) و69 حالة من المرضى شُخِصَ لديهم الإصابة بالابيضاضات اللمفاوية الحادة ALL بنسبة (48.9%) و5 حالات من المرضى شُخِصَ لديهم الإصابة بالابيضاضات الدموية الحادة ذات النمط الظاهري المختلط MPAL بنسبة (3.6%). كما اشارت نتائج الدراسة الحالية إلى أن نسبة المرضى الذكور المصابين بالابيضاضات الدموية اللمفاوية الحادة كانت حوالي (66.6%).
مجلة بحوث جامعة حلبRemoval of Some Chemical Contaminates (Sunset Yellow and Tartrazine) from Aqueous Solutions by Activated Carbon Prepareted from Peanut Shells
اُختبر في هذا البحث امتزاز صباغين هما التاارترازين (Tartrazine) ( TA ( وسنسات يلاو (Sunset Yellow) ( SY ( مان المحاليال المائياة دناد الدر اة 298K دلا ديناات الفحام الفعاال المنت اة والمحماري محليااو مان قشاور فساتق العبيد والمنشطة بحمض الفوسفور بنسب مختلفة ) 20-85wt% ( والمفحمة دناد الدر ة 450°C ، وكذلك ديناات منام منشاطة بحماض الفوسافور بنسابة 65wt% ومفحماااة دناااد در اااات حاااراري مختلفاااة ) 350-700C0 (. لاااوحن من منحنياااات الامتزاز لهذين الصباغين هاي مان النماوذل المنيماويرن ومن ساعة الامتازاز الأدنمي xm للسنست يلو تكون مكبر من التاارترازين، وتتعلاق ساعة الامتازاز الأدنمي بخواص الفحم )المسامية( وبطبيعة الصابا )ح مام - انحمليتام فاي الماااا (.
Research Journal of Aleppo University – Basic Science SeriesNew Simple Spectrophotometric Method for the Simultaneous Estimation of the Mixtures of Sildenafil and some Serotonin Reuptake Inhibitors
This work presents a simple, reproducible, sensitive, generic, and efficient method for simultaneous estimation. Vierordt’s method or simultaneous equation method was developed and validated for simultaneous estimation of two mixtures: Sildenafil and Duloxetine, Sildenafil and Fluoxetine, in the pure and pharmaceutical dosage forms. The method was based on the measurement of absorbance at two wavelengths for each mixture in methanol. Calibration curves of all drugs were found to be linear. With good correlation coefficient values of more than 0.99. The RSD values were found within limits and the recoveries ranged between 98 to 102%, confirming that the proposed method is accurate. This method for the simultaneous estimation of these four mixtures is simple and economical and can be applied successfully for the simultaneous 4 estimation of PDEs inhibitors and their combinations with some serotonin reuptake inhibitors in the pure and pharmaceuticals dosage forms.
Research journal of pharmacy and technologySpectrophotometric Method for Simultaneous Estimation of Dapoxetine and some Phosphodiesterase-5 inhibitors in new combinations
ABSTRACT: This work presents a simple, reproducible, sensitive, generic االنكليزية/ العربية بالغة الملخص and efficient method for simultaneous estimation. Vierordt’s method or simultaneous equation method was developed and validated for simultaneous estimation of two mixtures: Tadalafil and Dapoxetine, Vardenafil and Dapoxetine, in the pure and pharmaceutical dosage form. The method was based on the measurement of absorbance at two wavelengths for each mixture in methanol. Calibration curves of all drugs were found. with a good correlation coefficient value of more than 0.99. The RSD values were found within limits and the recoveries ranged from 98 to 102% which confirms that the proposed method is accurate. This method for simultaneous estimation is simple, and economical and can be applied successfully for the simultaneous estimation of Dapoxetine and its combinations with Tadalafil and Vardenafil in pure and pharmaceuticals dosage forms.
Research journal of pharmacy and technology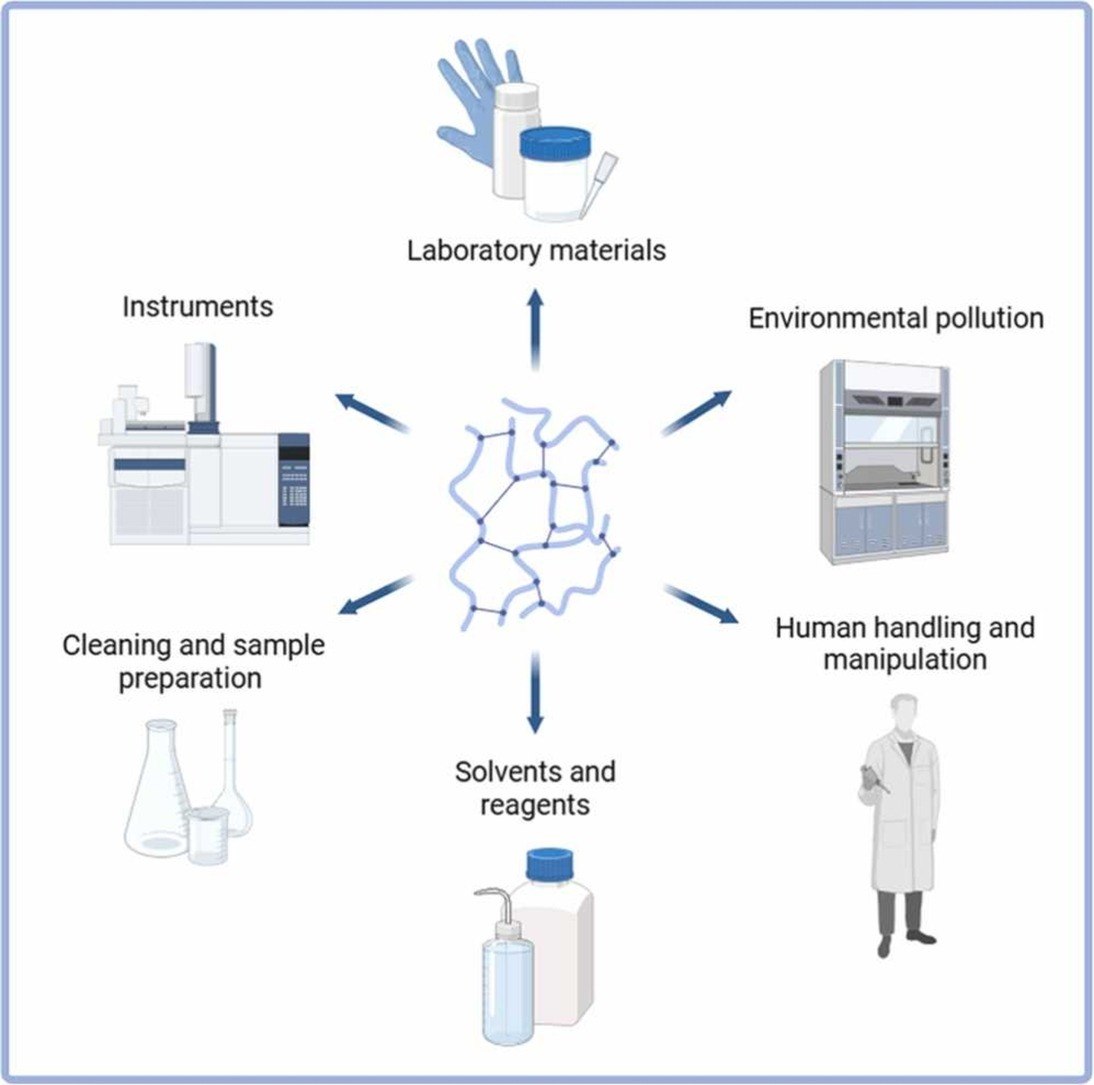
Cross-contamination pathways in the analysis of plastics and related chemical compounds: Good laboratory practices and tips
The analysis of plastics and related compounds at trace levels is highly susceptible to contamination because many target analytes are ubiquitous in laboratory environments. This work identifies six main contamination pathways: laboratory materials, environmental sources, human handling, solvents and reagents, cleaning and sample preparation, and instrumental systems. For each pathway, preventive strategies and good laboratory practices are proposed based on experimental experience and literature examples. The use of procedural blanks and early consideration of contamination risks during experimental design are strongly recommended. Overall, this study provides a structured framework to improve data reliability, reproducibility, and transparency in plastic-related contaminant analysis.
sciencedirect Journals
Sustainable harnessing of Red Sea brown seaweed extracts as plant growth promoters: Analytical profiling and application to faba bean
Seaweed liquid extracts (SLEs) are progressively recognized as sustainable biostimulants that enhance plant growth and stress resilience. This investigation evaluated the effects of SLEs derived from four seaweed species Cystoseira compressa, Sargassum aquifolium, Sargassum natans, and Turbinaria ornate on the seedling performance of Vicia faba L. Seedlings received weekly soaking applications of the extracts, which were chemically characterized for macro- and micronutrients, minerals, phenolics, and plant growth regulators. All SLE treatments significantly improved root development, esterase activity, and soluble sugar accumulation compared with controls (p ≤ 0.05). The extract of C. compressa, enriched in indole-3-acetic acid (IAA; 15 nM), displayed the strongest effect on root morphological traits. Extracts from S. aquifolium and S. natans, characterized by higher phenolic content, conspicuously enhanced plant nutritional status and the productivity of the plant, whereas T. ornata primarily stimulated root elongation without marked effects on nutrition. A clear relationship was perceived between extract composition and physiological response. These conclusions demonstrate the potential of seaweed based biostimulants to improve crop productivity through natural, resource-efficient inputs, contributing to sustainable agriculture and supporting the United Nations Sustainable Development Goals (SDGs). The valorization of marine biomass for agricultural use also reinforces blue economy strategies, particularly in the Red Sea region
sciencedirect JournalsAnalytical Green Star Area for Sample Preparation (AGSA-Prep) as a Comprehensive and Practice-Oriented Metric
Sample preparation is a critical, yet often environmentally burdensome, step in analytical chemistry. While Green Sample Preparation principles aim to mitigate this impact, existing assessment tools such as AGREEprep offer room for improvement in the assessment process. In particular, parameters such as material reusability, operator safety, and analytical performance constraints are not comprehensively considered, often resulting in lower scores for potentially green methods. This article presents AGSA-Prep (Analytical Green Star Area for Sample Preparation) as a new metric designed to provide a more comprehensive, balanced, and contextualized assessment of sample preparation greenness, seeking to supplement and update valid and currently accepted assessment tools. Building on the AGSA framework, AGSA-Prep incorporates additional assessment parameters, including reusability, waste management, energy consumption, automation, and operator protection based on personal protective equipment requirements. By simplifying complex scoring equations through categorical scoring systems, AGSA-Prep improves accessibility for routine use. This paper details the development of AGSA-Prep and critically compares its criteria with those of AGREE prep, demonstrating how AGSA-Prep offers a comprehensive assessment, thus guiding the development of greener, safer, and more efficient laboratory practices. The software is available for end-users at bit.ly/AGSA-Prep.
analytical science advances
Coupling MgSO4-assisted SALLE with a fluorimetric turn-off strategy for the determination of cinacalcet HCl in pharmaceutical and human matrices
An innovative, sustainable analytical approach was developed by coupling an Epsom salt, E518 (magnesium sulfate)–facilitated salting-out assisted liquid–liquid extraction (SALLE) system with a safe molecular fluorescent probe, Celfia Pink B (CPB), employing a fluorescence turn-off sensing strategy. MgSO₄ acts as a green salting-out agent, enhancing phase separation efficiency and producing cleaner extracts with higher analyte recovery. CPB, a safe, food-grade dye, serves as a selective fluorogenic probe whose emission at 553 nm is quenched upon electrostatic ion-pair complex formation with Cinacalcet HCl under mildly acidic conditions. The method exhibited excellent linearity (0.08–1.3 µg/mL) with high sensitivity (LOD = 0.024 µg/mL; LOQ = 0.075 µg/mL). Validation according to ICH guidelines demonstrated robustness, precision, and accuracy across pharmaceutical formulations and biological matrices. Sustainability assessment using the WAC framework categorized the platform as “white,” highlighting low environmental impact, safe operation, and simplicity. By minimizing organic solvent use and eliminating hazardous reagents, this MgSO₄-assisted SALLE/fluorescence turn-off approach provides a green, highly sensitive, and versatile tool for trace-level determination of Cinacalcet HCl in pharmaceutical and biological matrices.
Scientific Reports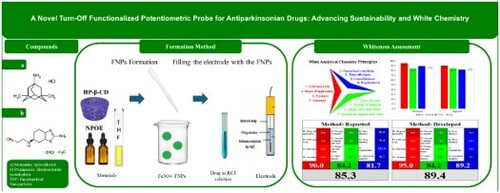
A novel turn-off functionalized potentiometric probe for antiparkinsonian drugs: advancing sustainability and white chemistry
To detect memantine hydrochloride (MEM) and pramipexole dihydrochloride monohydrate (PDM) in pure form, pharmaceutical dosage forms, and spiked human plasma with improved speed and sensitivity, this study presents a novel potentiometric strategy using functionalized magnetic nano-sized iron oxide particles (5 nm) with 2-hydroxypropyl-β-cyclodextrin (2HP-β-CD). These particles were incorporated into the internal solution of the sensor electrode. Performance surpassed previous sensors due to the unique properties of magnetic iron oxide, which enhanced sensitivity and selectivity. Electrochemical evaluation followed IUPAC standards. Linearity was observed for MEM and PDM within 1 × 10−7–1 × 10-² M & 1 × 10−8–1 × 10-² M, respectively. Six weeks of replicate calibration graphs confirmed long-term potential stability and repeatability. Accuracy was validated using the standard addition technique. Experimental variables such as plasticizer type, pH influence, temperature fluctuations, foreign substance interference, and nanoparticle concentration were optimized. This method reflects principles of sustainability by combining excellent analytical performance with environmental and economic benefits. The high whiteness score, assessed via the RGB12 tool, confirms its alignment with white chemistry. This work demonstrates that true sustainability requires not only green practices but also comprehensive attention to analytical efficiency, eco-friendliness, and affordability.
Green Analytical Chemistry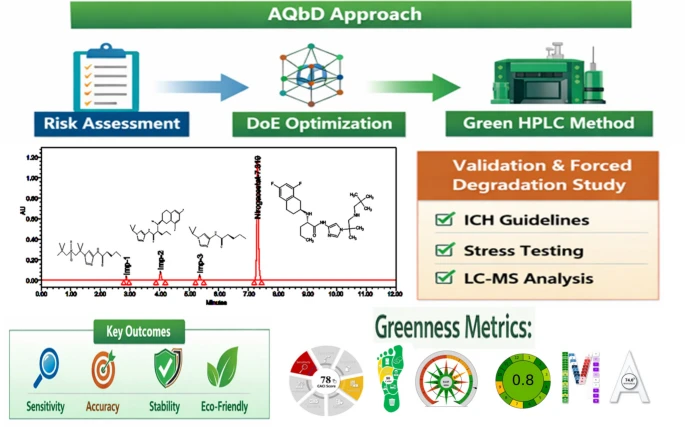
AQbD-driven development of a green RP-HPLC method for nirogacestat with LC–MS structural elucidation
Nirogacestat (NGT), a recently approved γ-secretase inhibitor for desmoid tumors, requires sensitive and sustainable analytical methods for impurity profiling. This study aimed to develop a green, stability-indicating HPLC method using an Analytical Quality by Design (AQbD) approach. Critical method parameters, including ethanol percentage, ammonium formate buffer concentration, and flow rate, were optimized through risk assessment and a central composite design to establish a robust design space. The final method employed an ethanol–ammonium formate mobile phase (24.14:75.86 v/v, 0.295 mM, pH 3.2) at 1.0 mL/min, achieving baseline separation of NGT, its process impurities, and degradation products with high efficiency (~ 15,900 plates) and symmetry (tailing factor ≤ 1.1). Validation following ICH Q2(R2) confirmed specificity, linearity (R2 > 0.999), accuracy (99.4–101.2% recovery), precision (%RSD ≤ 1.8%), and robustness. Sensitivity was demonstrated with LOD/LOQ values of 11.58/35.10 ppm for NGT and ≤ 0.52/1.58 ppm for impurities. Forced-degradation studies showed 3–14% degradation across stress conditions with mass balance between 95–105%, and peak-purity confirmed the stability-indicating capability. The method’s greenness was demonstrated using AGREE, AGSA, CACI, CaFRI, and a multi-assessment tool, with an overall whiteness index of 74%. Overall, this ethanol-based AQbD-optimized method offers a reliable, sustainable, and regulatory-compliant platform for impurity profiling and stability testing of NGT.
BMC chemistry journal
Systematic review of recent metrics (2020–2025) for greenness, applicability, and analytical performance with guidelines for practical use
In recent years, the demand for eco-friendly analytical methods with practical applicability and high analytical performance has driven the development of numerous evaluation metrics. This trend aligns with the principles of White Analytical Chemistry (WAC), which expands the scope of analytical science by integrating not only environmental friendliness and safety (green) but also analytical efficiency (red) and practical, economic aspects (blue). Over the last five years, several emerging tools have been introduced, offering more structured and systematic approaches for assessing analytical methods. This review classifies these tools into four main categories: (a) green tools, (b) blue tools, (c) red tools, and (d) multidimensional tools. The first parameter, such as reagent toxicity, solvent consumption, and waste generation, was identified as a key greenness indicator, representing the environmental component of broader analytical sustainability. The second addresses applicability, evaluating cost-effectiveness, time efficiency, and other practical aspects; the third concentrates on analytical performance, covering parameters essential for method validation. The fourth category integrates these aspects into comprehensive frameworks, enabling a balanced assessment across sustainability, applicability, and performance. This review critically compares these tools, outlining their merits and limitations. It also presents practical strategies to improve the greenness of conventional analytical methods. It proposes a novel set of standardized guidelines to ensure the transparent and non-manipulative application of sustainability metrics, addressing a critical gap in current practice. Additionally, our work discusses future directions toward comprehensive, objective, and universally adoptable assessment systems that can guide the next generation of sustainable analytical practices.
Results in Chemistry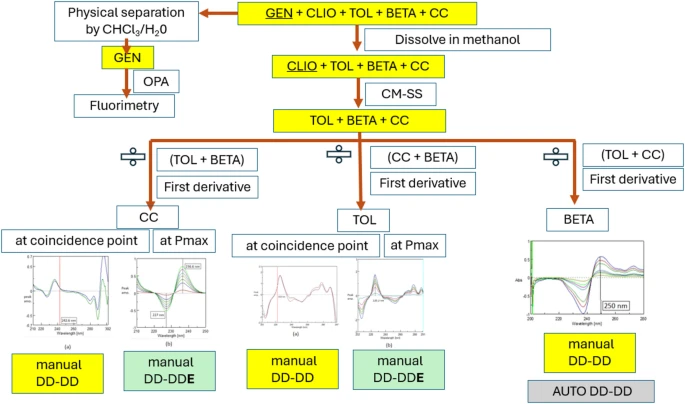
Artificial intelligence versus traditional approaches in multicomponent spectral analysis
This study explores the use of AI-assisted data handlingin spectrophotometric method development, providing a flexible and globally accessible alternative to traditional manual software algorithms.Quadriderm cream combines four active ingredients: Clioquinol (CLIO), Betamethasone (BETA), Tolnaftate (TOL), and Gentamicin (GEN) with the preservative Chlorocresol (CC). Building on our previous research on complex pharmaceutical mixtures with challenging ratios, this study applied established protocols for CLIO and GEN while focusing on the more analytically demanding ternary subsystem (TOL, BETA, and CC).The integration of AI-enhanced spectral handling and interpretation reduces operator-dependent variability and streamlines the analytical workflow. This includes generating calibration graphs and regression equations, as well as effectively handling scanned spectral data via consecutive prompts. Validation data such as accuracy and precision are assessed to ensure reliability. Furthermore, the system enables intelligent, simultaneous analysis of laboratory mixtures and pharmaceutical formulations, enhancing both efficiency and accuracy. The AI strategy, trained on spectral data supplied and monitored by the expertiseanalyst, can automatically predict optimal wavelengths with minimal interference, while manual handling strategy rely on analyst-driven selection. Two novel approaches were developed: the factorized derivative ratio extraction using double divisor (MAN-[DD- DDE])via Spectra Manager® software and the automated double divisor derivative ratio (AUTO-[DD-DD]) via AI tools and for resolving ternary mixtures with severely overlapping UV spectra and comparing the results with those of(MAN-[DD- DD])at coincidence points. Linear working ranges were 0.5–5.0 µg/mL (TOL), 3.0–30.0 µg/mL (BETA), and 2.0–20.0 µg/mL (CC); LODs were 0.09, 0.09, and 0.26 µg/mL, respectively. AI-driven data processing strategy matched the accuracy and reproducibility of traditional strategy manipulation while reducing subjective steps and effort. Finally, the UV-spectrophotometric method for pharmaceutical cream analysis was evaluated using the MA Tool (2025) to assess sustainability across green, white, and AI-driven criteria. AI-assisted scoring via Microsoft Copilot enabled rapid, reproducible assessment, yielding a Whiteness Score of 60.9% and providing actionable recommendations for greener and more efficient workflows
Scientific Reports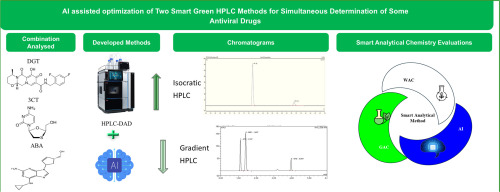
AI-assisted design and optimization of two smart green HPLC methods for simultaneous determination of some antiviral drugs
Two novel high-performance liquid chromatography (HPLC) methods were developed and validated for the determination of dolutegravir (DTG), lamivudine (3TC), and abacavir (ABA) in pharmaceutical formulations. In this study, AI engines such as Copilot, ChatGPT 5.2, Gemini, and Perplexity were integrated into HPLC method development. The selected predicted conditions underwent experimental optimization, validation, and refinement. Their practical implementation significantly improved efficiency and accuracy in the analytical process. The first method, an isocratic HPLC, was designed for the simultaneous quantification of DTG and 3TC using an XBridge® C18 column and a mobile phase of acetonitrile:phosphate buffer (pH 3.5, 50:50 v/v). Detection was performed at 258.0 nm for DTG and 275.0 nm for 3TC. The second method, a gradient HPLC, enabled the simultaneous quantification of DTG, 3TC, and ABA on a Spherisorb® ODS2 C18 column with methanol and TEA/TFA buffer (pH 3.15) as the mobile phase. Detection wavelengths were 258.0, 278.0, and 294.0 nm for DTG, 3TC, and ABA, respectively. The calibration ranges were wide and showed excellent linearity. Both techniques were validated in accordance with ICH Q2(R2) guidelines. The approaches align with smart analytical chemistry principles, combining green and white analytical chemistry (GAC and WAC) with AI-driven method development, resulting in accurate, fast, and sustainable techniques suitable for routine quality control applications.
Green Analytical Chemistry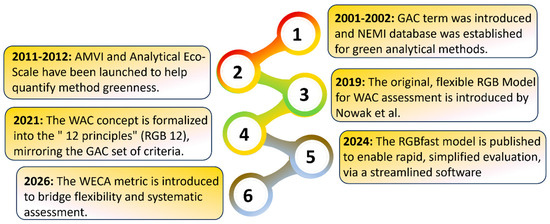
Whiteness Evaluation for Chemical Analysis (WECA) as a Flexible Tool and Web-Based Software for Whiteness Assessment in Analytical Chemistry
Two novel high-performance liquid chromatography (HPLC) methods were developed and validated for the determination of dolutegravir (DTG), lamivudine (3TC), and abacavir (ABA) in pharmaceutical formulations. In this study, AI engines such as Copilot, ChatGPT 5.2, Gemini, and Perplexity were integrated into HPLC method development. The selected predicted conditions underwent experimental optimization, validation, and refinement. Their practical implementation significantly improved efficiency and accuracy in the analytical process. The first method, an isocratic HPLC, was designed for the simultaneous quantification of DTG and 3TC using an XBridge® C18 column and a mobile phase of acetonitrile:phosphate buffer (pH 3.5, 50:50 v/v). Detection was performed at 258.0 nm for DTG and 275.0 nm for 3TC. The second method, a gradient HPLC, enabled the simultaneous quantification of DTG, 3TC, and ABA on a Spherisorb® ODS2 C18 column with methanol and TEA/TFA buffer (pH 3.15) as the mobile phase. Detection wavelengths were 258.0, 278.0, and 294.0 nm for DTG, 3TC, and ABA, respectively. The calibration ranges were wide and showed excellent linearity. Both techniques were validated in accordance with ICH Q2(R2) guidelines. The approaches align with smart analytical chemistry principles, combining green and white analytical chemistry (GAC and WAC) with AI-driven method development, resulting in accurate, fast, and sustainable techniques suitable for routine quality control applications.
AnalyticaTotal Synthesis of Cyano-3-Ethoxycarbonyl-4-Methyl-7-Phthalimido-7Δ-Cepheme: Stereochemical Study
الاصطناع الكلي لمركب سيانو-3-إيثوكسي كاربونيل-4-ميثيل-7-فثاليميدو-7Δ-سيفيم: دراسة استريوشيمية
Sulfur Letters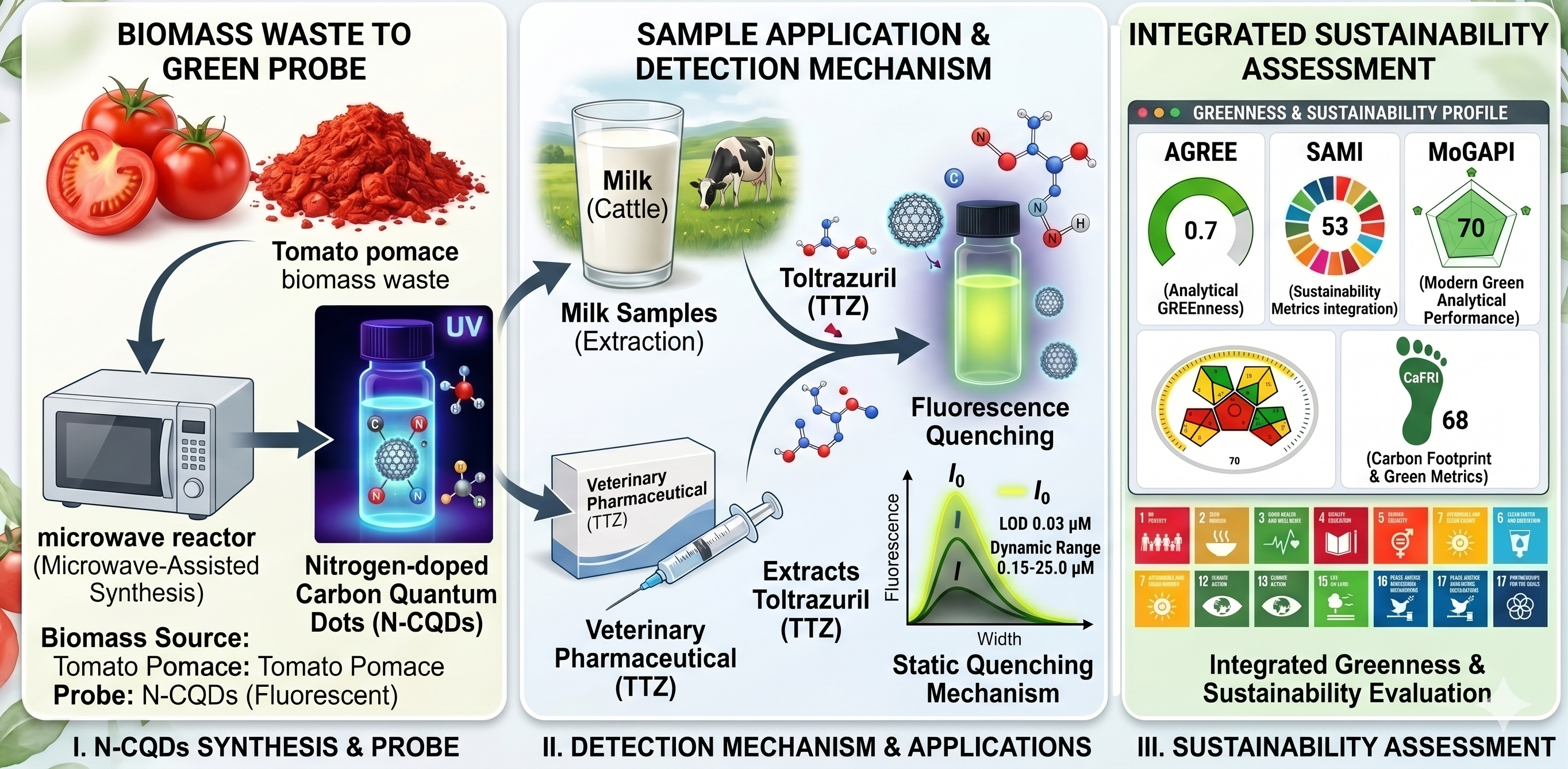
Utilization of Tomato Waste Biomass as a Fluorescent Sensor for Quantification and Residual Determination of Toltrazuril in Veterinary Drugs and Milk Samples
This study focuses on developing an analytical method for the determination of toltrazuril (TTZ), an antiparasitic drug widely used in veterinary medicine, particularly for protecting poultry and livestock from protozoal infections in hot and humid environmental conditions. Methodology: A fluorescent probe based on nitrogen-doped carbon quantum dots (N-CQDs) was prepared. These nanodots were synthesized using a domestic microwave-assisted method, utilizing tomato waste residues as a sustainable and low-cost biomass source. The prepared N-CQDs were applied for the first time as a fluorescent sensor for detecting TTZ in pharmaceutical formulations and milk samples. Key Results: 1. Detection mechanism: The method is based on static fluorescence quenching and demonstrated high sensitivity with a limit of detection (LOD) of 0.03 μM, within a linear range of 0.15 to 25.0 μM. 2. Practical application: The results confirmed the probe’s efficiency in monitoring food safety of animal origin and ensuring the absence of drug residues. 3. Extraction: A standardized and efficient extraction protocol was developed for isolating TTZ from cow milk samples to ensure measurement accuracy. Sustainability assessment: The proposed method was evaluated using modern green chemistry metrics such as AGREE and MoGAPI, as well as carbon footprint indicators including CaFRI, EPPI, and SAMI. The assessment confirmed that the method is not only efficient but also highly sustainable and environmentally friendly.
BMC CHEMISTRY