AQbD-driven development of a green RP-HPLC method for nirogacestat with LC–MS structural elucidation
- Publish Date: 10 - February - 2026
Research Abstract:
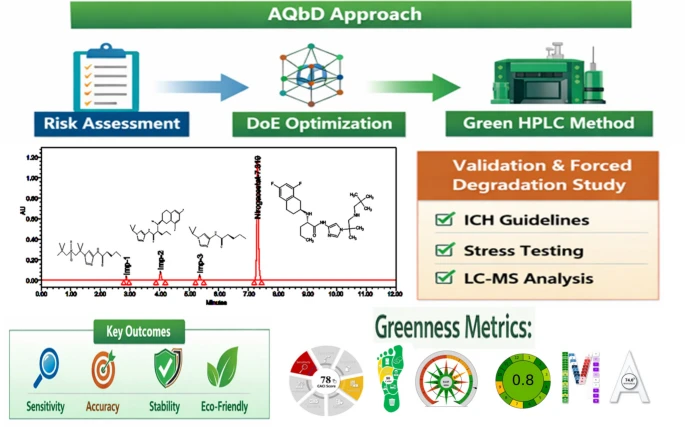
Nirogacestat (NGT), a recently approved γ-secretase inhibitor for desmoid tumors, requires sensitive and sustainable analytical methods for impurity profiling. This study aimed to develop a green, stability-indicating HPLC method using an Analytical Quality by Design (AQbD) approach. Critical method parameters, including ethanol percentage, ammonium formate buffer concentration, and flow rate, were optimized through risk assessment and a central composite design to establish a robust design space. The final method employed an ethanol–ammonium formate mobile phase (24.14:75.86 v/v, 0.295 mM, pH 3.2) at 1.0 mL/min, achieving baseline separation of NGT, its process impurities, and degradation products with high efficiency (~ 15,900 plates) and symmetry (tailing factor ≤ 1.1). Validation following ICH Q2(R2) confirmed specificity, linearity (R2 > 0.999), accuracy (99.4–101.2% recovery), precision (%RSD ≤ 1.8%), and robustness. Sensitivity was demonstrated with LOD/LOQ values of 11.58/35.10 ppm for NGT and ≤ 0.52/1.58 ppm for impurities. Forced-degradation studies showed 3–14% degradation across stress conditions with mass balance between 95–105%, and peak-purity confirmed the stability-indicating capability. The method’s greenness was demonstrated using AGREE, AGSA, CACI, CaFRI, and a multi-assessment tool, with an overall whiteness index of 74%. Overall, this ethanol-based AQbD-optimized method offers a reliable, sustainable, and regulatory-compliant platform for impurity profiling and stability testing of NGT.
Publishers:
